- ABOUT JNU
- ADMISSION
-
ACADEMICS
- Schools and Colleges
-
Departments and Programs
- Arts College of
- Chinese Language and Culture College of
- Economics College of
- Electrical and Information Engineering College of
- Foreign Studies College of
- Information Science and Technology College of
- Environment School of
- Humanities School of
- International Business School
- International Studies School of
- Journalism and Communication College of
- Law School
- Liberal Arts College of
- Life Science and Technology College of
- Management School of
- Marxism School of
- Medicine School of
- Pharmacy College of
- Physical Education School of
- Science and Engineering College of
- Shenzhen Tourism College
- Research Institute
- Research Center
- Programs in English
- Majors
- Study Abroad
- Online Learning
- RESEARCH
- CAMPUS LIFE
- JOIN US
Palladium Nanoclusters Protected by Carbonyl Metallates Enable Visible-Light-Driven Carbonylation Using Atmospheric CO
Recently, the research team led by Professors Dan Li, Tao Wu, and Shangfu Yuan from the College of Chemistry and Materials Science at Jinan University published a study titled "Carbonylmetallated Palladium Nanoclusters for Visible-Light-Driven Carbonylation under 1 atm of CO" in the Journal of the American Chemical Society, an internationally authoritative academic journal.
Palladium metal plays a crucial role in organic synthesis and energy conversion due to its exceptional catalytic activity. However, compared to extensively studied coinage metal nanoclusters, reported examples of palladium nanoclusters remain scarce. Early reports of palladium nanoclusters often suffered from poor stability or solubility, involved cumbersome synthesis procedures, and required inert gas protection. Although direct reduction is a common method for preparing gold or silver nanoclusters, it is unsuitable for palladium nanoclusters due to the rapid reduction kinetics of palladium ions, which tends to form large nanoparticles and offers poor synthetic controllability. To date, only a few low-nuclearity Pd(0) nanoclusters have been successfully prepared via reduction, and even these were obtained in very low yields.
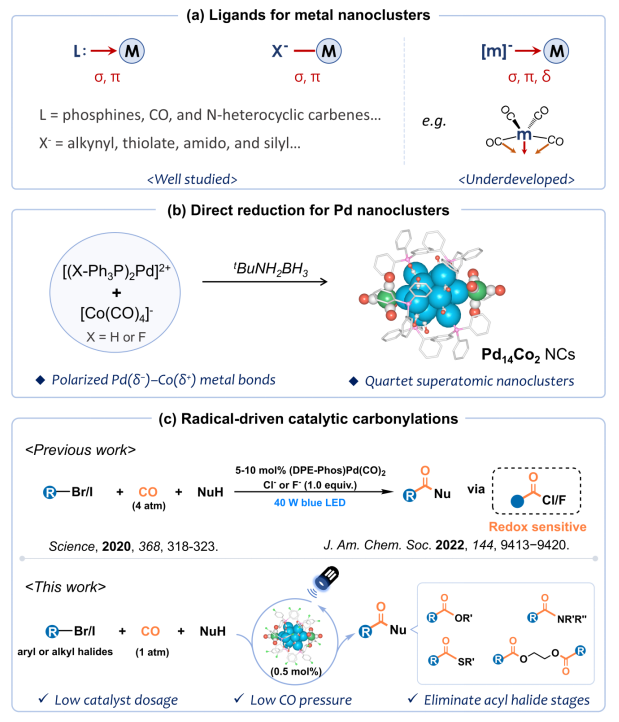
The key to achieving direct reductive synthesis of palladium nanoclusters lies in effectively controlling the reduction kinetics of palladium ions. Introducing appropriate protective agents during synthesis can suppress the reduction rate, stabilize the clusters, and prevent their aggregation. Zero-valent palladium with a d¹⁰ electronic configuration exhibits weak Pd–Pd metal bonding and a relatively low cohesion energy (377 kJ mol⁻¹). Its stability relies on protection by strong σ-donor/π-acceptor ligands. Carbonyl metallate anions, a class of transition metal-based ligands ([M(CO)ₓ]⁻), act as Lewis bases to donate electrons to metal ions and have been widely studied in organometallic and cluster chemistry. These metal ligands feature both metal coordination sites and CO π-coordinating groups, enabling diverse multiple metal-metal bonding interactions with palladium centers, making them ideal protective agents for modulating palladium reduction kinetics and achieving controlled synthesis.
In light of this, the research team of Professors Shangfu Yuan, Tao Wu, and Dan Li from Jinan University, in collaboration with Associate Professor Jianyu Wei from Ningxia University, proposed an innovative synthesis strategy utilizing carbonyl metallate ligands to regulate palladium reduction kinetics. They successfully constructed Pd₁₄ nanoclusters exhibiting "open-shell superatom" characteristics, protected by [Co(CO)₄]⁻ anionic ligands, and applied them to visible-light-driven carbonylation reactions of organic substrates using atmospheric CO.
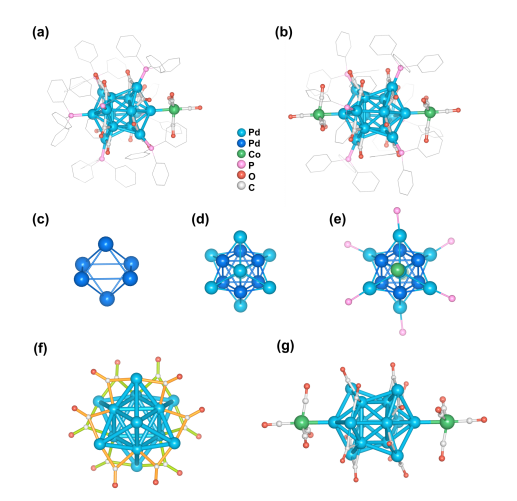
Single-crystal X-ray diffraction analysis revealed that the Pd₁₄ nanocluster core features an octa-capped octahedral geometry. Two [Co(CO)₄]⁻ ligands are anchored at opposite ends of the Pd₁₄ core via apical coordination modes. Combined characterization by ESI-MS, XPS, and SQUID, corroborated by DFT theoretical calculations, confirmed that the Pd₁₄ cluster exists in a quartet ground state and exhibits rare open-shell superatomic character, following the jellium electron configuration 1S¹. Furthermore, valence bond structure analysis indicated the presence of polar Pd(δ⁻)–Co(δ⁺) bonds on the cluster surface, alongside multiple orbital interactions between the [Co(CO)₄]⁻ ligands and the Pd₁₄ metal core. This CO-assisted bimetallic bonding mode confers excellent stability to the cluster.
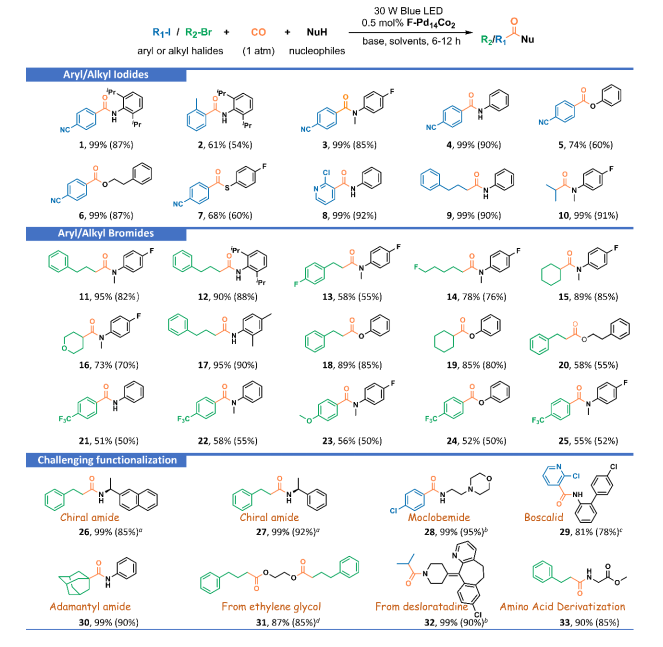
Notably, the F-Pd₁₄Co₂ nanocluster demonstrated exceptional catalytic activity for carbonylation reactions under visible light irradiation. Under mild conditions of 1 atm CO and a 30 W 450 nm LED lamp, this catalytic system efficiently coupled various challenging aryl and alkyl halides with inert nucleophiles, producing over 30 high-value carbonyl derivatives, including amides, esters, and thioesters. Additionally, the catalyst proved effective for the derivatization of complex natural molecules and the targeted synthesis of bioactive pharmaceuticals, such as the monoamine oxidase inhibitor moclobemide and the antifungal agent boscalid. Mechanistic studies suggested that the catalytic process involves metal-cooperative photoinduced single electron transfer. The reaction pathway bypasses the formation of acyl halide intermediates, attributed to the terminal [Co(CO)₄]⁻ groups on the palladium nanoclusters, which assist in activating nucleophiles like amines and alcohols while lowering the energy barrier for nucleophilic attack.
This work validates the effectiveness of carbonyl metallate ligands in controlling palladium reduction kinetics, enabling the controlled synthesis of palladium nanoclusters with polar heterometallic interfaces. Upon visible-light excitation, these heterometallic palladium clusters facilitate efficient carbonylation of organic substrate molecules under atmospheric CO pressure, synthesizing a series of high-value carbonyl derivatives like amides, esters, and thioesters. This study proposes a novel synthetic pathway for creating new palladium nanoclusters and offers a fresh construction strategy for designing polar heterometallic nanoclusters. It holds promise for expanding the types of multicomponent cross-coupling catalytic reactions utilizing novel palladium nanocluster catalysts.
The research was recently published in the Journal of the American Chemical Society. Doctoral student Linmei Zhang and Master's student Huizhi Wei are co-first authors. The corresponding authors are Professors Shangfu Yuan, Tao Wu, and Dan Li from the College of Chemistry and Materials Science, Jinan University, and Associate Professor Jianyu Wei from Ningxia University.
Link to the paper: https://pubs.acs.org/doi/10.1021/jacs.5c08034
NEWS
- About the University
- Quick Links
Copyright © 2016 Jinan University. All Rights Reserved.




